White blood cells
White blood cells are a diverse group of cells, originating from different precursor cells. Although the specific functions of the white blood cell subtypes differ widely, all of them exist to defend the organism against non-endogenous agents in the broadest sense. Myeloid white blood cells are produced in bone marrow. Lymphoid cell development, however, is not restricted to bone marrow and takes place in primary and secondary lymphatic organs.
The total number of white blood cells in healthy adults ranges approximately between 4‒10 x106/µL, of which neutrophils and lymphocytes make up the majority. Monocytes, eosinophils and basophils are found in far smaller numbers in healthy persons. The absolute count of each white blood cell subtype in peripheral blood has clinical significance and is more informative than the relative count given as a percentage. White blood cell pathologies affecting either the myeloid or lymphoid lineage can be the result of both reactive and non-reactive (malignant) disease. Reactive changes are observed in the course of infectious or inflammatory diseases whereas malignant alterations point to leukaemia, lymphomas and other haematologic malignancies.
To distinguish between different diseases related to white blood cells, determining both their number and their exact type and state of maturity and activation is crucial. Automated haematology analysis is an essential component in the diagnostic process and helps to identify the presence of disease by providing accurate cell counts, highlighting conspicuous cell populations and providing additional information on the cell functionality, e.g. for neutrophil and lymphocyte activation. With white blood cell diseases, finding the right diagnosis is complex and needs all the information available from the complete blood count, morphology analysis, immune phenotyping, and other tests.
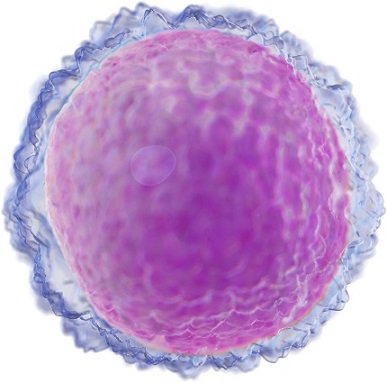
Lymphocytes defend the organism against infection by distinguishing the body’s own cells from foreign ones. Molecules recognised by the body as foreign are known as antigens. Each lymphocyte is only stimulated by one specific antigen. When lymphocytes recognise this antigen, they produce chemicals to fight it.
There are three main types of lymphocytes: B cells, T cells and natural killer cells. Although ‒ compared with other white blood cells ‒ all lymphocytes are small and round without granules, there is a large variety of different subtypes, and distinguishing between them morphologically is tricky.
Reasons for an increased lymphocyte count include infection or inflammation, as well as certain types of malignancies, especially haematologic malignancies. In addition to giving an absolute and relative lymphocyte count, several flags on Sysmex analysers can point to suspicious lymphocytes for which presence a follow-up test should be performed.
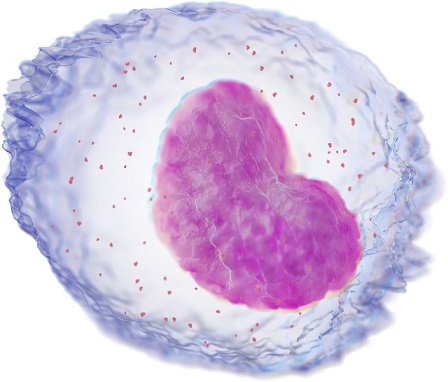
Monocytes play a key role in the immune response. They can quickly move to infection sites and differentiate into macrophages and dendritic cells to provoke an immune response. Cells of the monocyte-macrophage system can engulf foreign particles and break them down to antigens, which they can then present on their surface.
Automated monocyte counts are available as the ratio of monocytes to the total count of white blood cells, or as an absolute count. An increased monocyte count may be indicative of various disease states, e.g. chronic inflammation or infection, but may also occur in malignant diseases such as chronic myelomonocytic leukaemia.
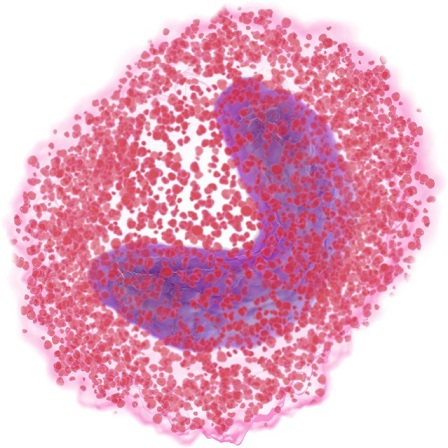
Like neutrophils, eosinophils belong to the granulocytes because they are filled with granules containing different enzymes. Eosinophils are best known for their roles in anti-helminth immunity and allergic diseases including those affecting the skin, respiratory system and gastrointestinal tract. They can also move and phagocytose particles. Since they kill helminthic parasites by releasing certain cytotoxic enzymes to amplify the inflammatory response and are involved in hypersensitivity reactions, an increased eosinophil count is most likely associated with parasite infestation or allergy. However, eosinophilia can also point to malignant diseases as it is evident in some types of neoplasia.
Basophils are the white blood cells least represented in peripheral blood; they also belong to the granulocytes. Like with eosinophils, an increase in the basophil count often ‒ though not always ‒ points to allergy or parasitic infections. Basophils are also present at the site of an inflammatory reaction. They seem to play a role similar to that of eosinophils in the defence against pathogens. Like eosinophils, they are recruited to the sites of allergic reactions and function together with mast cells as effector cells in complex processes such as chemotaxis or cell adhesion and fulfil an immunomodulatory role during allergic reactions.
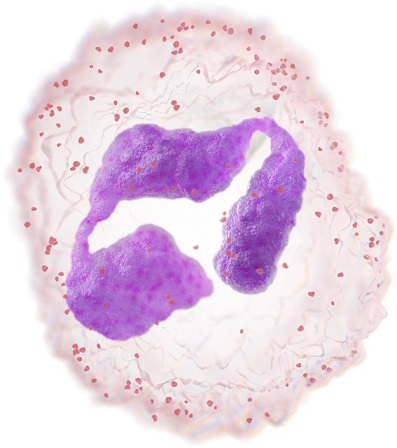
Neutrophils belong to the granulocytes, which are characterised by the presence of granules in their cytoplasm. They play an important role in immune defence and are the first immune cells to arrive at a site of inflammation – usually within minutes. This happens through a process known as ‘chemotaxis’. Neutrophils can phagocytose other cells which seem harmful to the organism, such as bacteria. Yet they will not survive this act: pus consists mainly of dead neutrophils and digested bacteria.
Acute infections commonly cause various degrees of neutrophilia due to the release of segmented neutrophils and bands from the bone marrow and marginating pool. This reactive neutrophilia is often associated with the presence of toxic granulation, Doehle bodies, cytoplasmic vacuoles and elevated levels of leucocyte (neutrophil) alkaline phosphatase (LAP).
An increased neutrophil count can also be found due to primary abnormalities in the regulation of bone marrow neutrophil production, or physiologically in non-pathological situations, e.g. after stress or in smokers.
The absolute and relative neutrophil count can provide some information for diagnosis and monitoring of infections and is also watched during chemotherapy.